KIET School of Pharmacy
M Pharm in Pharmaceutical Quality Assurance Quality Assurance is two year postgraduation course that covers the latest industry trends, standards like ISO and Six Sigma, and the importance of quality assurance in the manufacture of pharmaceuticals. Quality Assurance is a vital component of the pharmaceutical industry, performed with the aim of providing superior quality end products. Various aspects of Quality Assurance, documentation, regulatory affairs, training, and analysis on advanced equipment are included in the course’s curriculum.
Pharmaceutical Quality Assurance department in KSOP is approved by regulatory bodies PCI, AICTE and AKTU (Lucknow). The department is well equipped with all necessary equipment like Gas Chromatography, High-Performance Liquid Chromatography, UV Spectrophotometry, FT-IR and many other analytical types of equipment. The department also has well qualified and experienced faculty members in their respective fields. This two year program is starting from 2019-20.
Career Opportunities after M. Pharm. in Pharmaceutical Quality Assurance
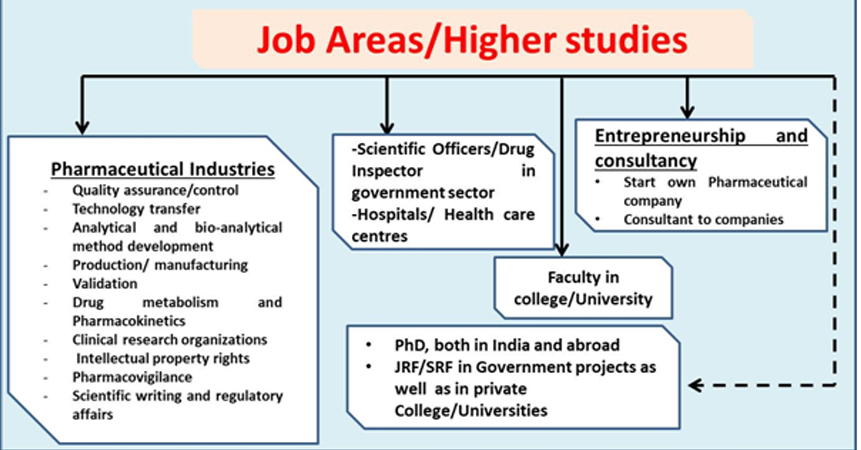
After M. Pharm in Pharmaceutical Quality Assurance students have job opportunities in vast areas such as:
- Pharmaceutical Industries: There are different areas such as quality assurance/control, technology transfer, clinical research organizations, property rights, analytical and bio-analytical method development, pharmacovigilance, scientific writing, production/ manufacturing, validation aspects, drug metabolism and Pharmacokinetics (DMPK) and regulatory affairs where candidates of both the disciplines are becoming recruited.
- Entrepreneurship and consultancy: Post graduates students are also able to train and encouraged to create their career as entrepreneurs to start their own company or as consultants to pharmaceutical companies.
- Higher studies and academics: student can also pursue PhD in further research and also for teaching jobs in academic institutions and as JRF/SRF in research projects.